bacti barrier: Active Defense Against Microbes
Download the original article by clicking here
The Problem
Hl Hl ... MRSA ... E. coli ... Salmonella ... Mold ... Allergens ... The world we live in is a dangerous place. The media tells an ominous story that threatens us all -the story of drug-resistant microbes and global pandemics. You take every precaution: you wash your hands and are careful about what you touch. Still, you can't help feeling vulnerable.
Chemists have responded with a dizzying array of products to destroy germs. Most of these antimicrobials use metals or poisons that leach into the surrounding environment to kill offending organisms. These agents create a halo of diminishing efficacy around the area of application. Microbes near the center of this halo are usually eliminated, but those on the outer edge, where the strength of the poison is greatly reduced, survive and mutate into resistant strains, known as "Super Bugs".
Scientists and medical professionals see the real danger: most antimicrobials actually make the problem worse by promoting adaptive strains of microorganisms.
The Solution
The answer? The BactiBarrier Cleaning System. The BactiBarrier" cleaning system approaches cleaning in a more logical way. We have achieved groundbreaking success using our two-step cleaning system.
1. First we remove and neutralize unwanted microbes by cleaning away pollutants using our premium EPA registered Disinfectant/Cleaner antimicrobial.
2. Second we apply an odorless, bound, EPA-registered, antimicrobial barrier of protection on surfaces that may be exposed to futureharmful microbial growth.
This two-part cleaning and protection system is a unique combination of both innovative procedures and chemistry. Efficient and effective cleaning procedures require careful attention to details. Proper application, coverage and dwell time of our Step 1 BactiBarrier Disinfectant/Cleaner antimicrobial will eliminate 99.9 o/o of the unwanted microbial growth that builds up on a hard
surface.
BactiBarrier Protector or Step 2, applied properly, provides a bonded, long-term, antimicrobial protective barrier. This chemistry works slightly different than the first step. Its unique charged mechanical action actually works to eliminate microbial growth for months or even years on/in specific undisturbed hard and soft surfaces.
How the BactiBarrier Cleaning System Works
At the heart of our process is a molecule made of a long chain of atoms, forming a microscopic "spear". The silane base ofthis spear forms a covalent bond with virtually any surface and locks it, in turn, to each adjoining molecule. Built into the spear's shaft is a positively charged nitrogen atom. This positive charge irresistibly draws negatively charged, airborne microbes from the atmosphere, impaling them on the point of the spear. As the attraction pulls the microbe farther down the spike, they contact the positively charged nitrogen atom and are destroyed by its minute electrical discharge.
Since microbes are obliterated instead of being altered, they can not learn to adapt to this electro-mechanical process. In over thirty years of field application, no adaptive strains have ever been documented. Best of all, because nothing of essence is transmitted during the process, the efficacy of our barrier does not diminish- it keeps on working, month after month.
Now, imagine an entire field of these electrically charged spears- billions upon billions of them- bonded to a surface and interlocked one to another, points at the ready. This is the inhospitable bio-barrier that confronts any bacteria, virus, or fungi that ventures near the surface - our active defense against microbes.
Benefits of the BactiBarrier Cleaning System
- Proven BactiBarrier active bio-barrier
- Ongoing protection that lasts month after month
- Covalently bonds to almost any surface
- EPA registered & FDA approved products
- Effective against a broad spectrum of gram(+) & gram(-)
- bacteria, viruses, & protozoa, including mold, mildew, & algae
- Extensively field-tested for years in hundreds of products & locations
- Environmentally friendly, non-toxic, non-leaching products
- No noxious poisons, heavy metals, or harsh fumes
- Electro-mechanical action does not permit adaptive strains
- Uniform coating does not alter the texture of surfaces
- Reduces airborne contaminants while inhibiting surface growth
- Not affected by normal housekeeping procedures, including steam cleaning
- Presence & efficacy can be verified with a simple field test
- Minimizes institutional liability by demonstrating a proact!ve approach
- Long-term efficacy saves money over temporary treatments
Some examples of potential applications:
Healthcare, building and remodeling, athletic facilities, transportation, education, day-care, hotels and restaurants, correctional facilities, cruise lines, residential, business, marine, medical devices, industry and government, etc. This is just a small sampling of where BactiBarrier can be applied.
The comprehensive BactiBarrier Cleaning System can dramatically reduce the bio-load on touch surfaces, while lowering airborne contaminants in the process. BactiBarrier creates a biological safety net over your entire environment, protecting you from pathogenic organisms and fungi, month after month.
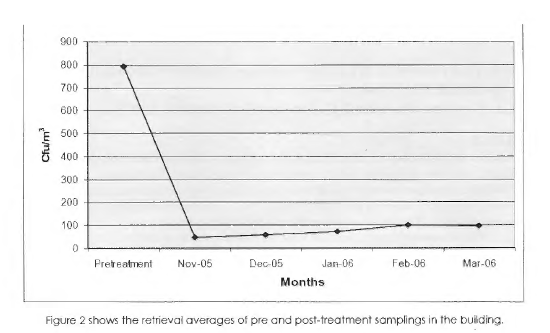
The research rooms were in the Swan Hotel. Room 409 was cleaned by housekeeping staff and was unoccupied at the time of the research. Room 457 was not cleaned and was unoccupied at the time of the research. Moisture readings were within normal limits on structural materials at the time of the research. Walls, ceilings, bathroom fixtures and contents (e.g., telephone, remote control, thermostat, upholstery, etc.) were cleaned followed by an electrostatic application of BactiBarrier™.
The self-contained HV AC units were sealed at the time of cleaning and application of BactiBarrier™. Pre-cleaning samples were collected and following cleaning and treatment post-cleaning/treatment samples were collected.
Sampling Procedures
The information and results collected during this study have been divided into the following categories: Visual Inspection, odors, and Microbiology.
Visual Inspection: A limited visual assessment was conducted throughout the units. Units were reasonably clean and consistent with occupancy.
Odor: Observations were made for detectable odors.
A musty, earthy moldy odor was apparent on entering each room.
Microbiology: Microbiological sampling to measure microbial levels on surface and air samples were collected out of doors and inside the units and swab and dust samples were collected from various surfaces (e.g., countertops, bath fixtures, telephone, remote control, carpet and upholstery).
Microbiological Testing
When collecting samples, this represents the environmental conditions that are present at the time qf sampling. It is a snapshot of the current conditions, which change with the time of day, amount of indoor activity, and weather conditions.
10 Adenosine triphosphate (A TP) samples were collected. A TP measures the biological activity on surfaces (bacteria, mold and other biological contamination). The counts are measured in relative light units (RLU).
In room 409 (Cleaned-Vacant):


Results:
Odors were reduced significantly after cleaning and application. Based on the data reported, the cleaning and BactiBarrier™ application compare favorably to other similar indoor environments and reduced microbial burden. The microbial reduction observed was substantial.
Thank you for allowing me the opportunity to research this new technology that can be applied to material and surfaces, reducing contaminants and making the surface antimicrobially active. If you have any questions or require additional information, please do not hesitate to give me a call.
Respectfully submitted,
Daniel Bernazzani,
PhD Candidate Environmental Studies
Antioch University New England
225 Humphrey St., #3
Swampscott, MA 01907
Master Restorer- IICRC
Applied Microbial Remediation Specialist- IICRC
Enclosures: Laboratory Analytical Data Sheets
Improved Control of Microbial Exposure Hazards in Hospitals:
A 30-Month Field Study
R.A. Kemperl, L. Ayers2, C. Jacobson3, C. Smith4, and W.C. WhiteS
Abstract
The microbial colonization of environmental surfaces in hospitals and other buildings can produce infective, allergenic and toxigenic risks for occupants. Traditional disinfectant/sanitizer formulations do not provide sustained control of microbial contamination at low levels and their extended use is potentially dangerous to man and the environment. This study evaluates the effectiveness of a new class of antimicrobial agents that covalently bond to surfaces and are not chemically reactive with the microbial cells. This organosilicon antimicrobial, 3-trimethoxysilylpropyldimethyloctadecyl ammonium chloride (BactiBarrier® Antimicrobial), produces antimicrobially active surfaces on a variety of substrates. After modifying the interior surfaces of a flooddamaged hospital with this antimicrobial, we evaluated airborne microbial concentrations for 30 months. The results show a significant and sustained reduction of viable airborne microorganisms.
Conclusions:
The data from this study show that significant control of airborne microorganisms results from the modification or interior building surfaces with an organosiliconantimicrobial. Even when evaluated under severe environmental conditions, the antimicrobial activity of these modified surfaces provides substantive reduction ofairborne microbial concentration. The initial reduction of airborne microorganisms and the sustained control of microbial levels during the 30 months of this study are unprecedented in the literature. When viewed collectively, the safety, efficacy, and durability of this 9h 03/2005 Hospital Study technology provide a unique opportunity to control the risks associated with microbial contamination in buildings.
Measurement of Cleaning Effectiveness in School Buildings:
Evaluation of a Candidate Standard Method
Eugene C. Cole, DrPH1
, Richard Shaughnessy, PhD2
, Ulla Haverinen-Shaughnessl
PhD, Keith E. Leese4
Cleaning procedure on school desks:
A cleaning protocol combined initial dry wiping with microfiber cloth, followed by spray disinfectant-cleaner, and a final microfiber wiping until dry. Surfaces were measured before and after cleaning by three commercially available and widely used ATP systems in each school using a swabbing technique and 25 cm2 template. Each school was sampled three times per week - each time with a different A TP system. Sampling included 5 desktops per 5th grade classroom (2 classrooms per school). Each desktop was marked to assure it was sampled on each day; and the areas sampled were different on each sampling day to avoid re-sampling of the same areas. Similarly, 5 cafeteria tables were also similarly sampled on each of the 3 sampling days using the same approach. Sampling of restroom surfaces included 10 interior stall door surfaces and 10 sink side areas. Samples from each surface/site were also taken for total culturable bacteria using surface contact agar plates as a measure of microbial reduction and improved hygiene.
Conclusion:
Specific ATP values/ranges appear to be a function of both surface material type and location within a school building, and further study is expected to confirm unique values for such critical compartments of the school building ecosystem. It is anticipated that such "normal" ranges can be established for each surface type, and that these can be used in the routine monitoring of cleaning effectiveness in school buildings.
5 Acknowledgements
This study was co-sponsored by the Cleaning Industry Research Institute (CIRI) and the ISSA - The Worldwide Cleaning Industry Association. The authors thank Alesia Bailey of the University of Tulsa, and the administrators and staff of the participating schools.
Study 3
Reducing Microbial Contamination in Hospital Blankets
By James W. Krueger
NOSOCOMIAL INFECTION
Nosocomial infection is a serious issue for health care facilities. According to recent articles, 1.8 million Americans contract nosocomial infection from hospitals every year. 20,000 patients died in 1998 as a direct result of nosocomial infection and 70,000 die from complications caused by infection. The cost of treating nosocomial infection in the United States is estimated at $4.5 billion a year. Controlling infection in an environment which is contaminated by the nature of its function is difficult at best and requires a multifaceted approach.
1. Will hospital blankets, protected by the BactiBarrier Protector technology, end nosocomial infections? No.
2. Will hospital blankets, protected by the BactiBarrier Protector technology, reduce bacterial and fungal contamination on the blanket? Yes.
3. Will the use of hospital blankets, protected by the BactiBarrier Protector technology, be a positive step toward asepsis in the patient=s immediate environment? Yes.
4. Is the use of hospital blankets, protected by the BactiBarrier Protector technology, a component of practicing reasonable care? Yes.
Summary:
The In-use study on Spartan Mills blankets correlates well with the simulated study undertaken earlier in the year. Both studies clearly show that blankets protected by the BactiBarrier Protector technology have a significantly lower bioburden and will present less of a risk in the patient environment. Historical data generated by American Hospital Supply and Dow Corning Corporation support these findings. These data generated by university, medical and industrial laboratories represent some of the most extensive microbiological work ever performed on antimicrobial treated substrates for use in the medical community. The control of the microorganisms is impressive and provides numerous benefits.
Prevents blanket staining due to mold and mildew growth that occurs on damp blankets prior to laundering.
- Controls blanket deterioration due to microbial growth that occurs on blankets during storage.
- Controls odors caused by bacteria and fungus normally found in blankets.
- Provides 3 times more protection from bacteria and fungus than an untreated blanket.
See full Study www.bactibarrier.com/studies